3Hepatitis C drug therapy. EASL recommendations on treatment of hepatitis C.

Residual Risk Of Liver Disease After Hepatitis C Virus Eradication Journal Of Hepatology
Thomas3 and Leonard B.
Hepatitis c guidelines pdf. Guidelines for the screening care and treatment of persons with hepatitis C infection. Numerous guidelines produced annually by prestigious medical bodies outline gold standard practices and are aimed at physicians in resource-rich environments. Chronic hepatitis C can be made after this time period.
Case is a suspected re-infection. The guidelines for post-exposure prophylaxis are updated. 1Hepatitis C prevention and control.
In the case of suspected acute hepatitis C in immuno-compromised patients and in. Before hepatitis symptom onset until two weeks after symptom onset. Numerous guidelines produced annually by prestigious medical bodies outline gold standard practices and are aimed at physicians in resource-rich environments.
Complete the Hepatitis C perinatal form httpswwwdohwagovPortals1DocumentsPubs150-113-ReportForm-HepC-Perinatalpdf and enter the data into WDRS using the Perinatal HCV wizard. Final update of the seriesq European Association for the Study of the Liver Summary Hepatitis C virus HCV infection is a major cause of chronic liver disease with approximately 71 million chronically infected in-dividuals worldwide. The infectious period time period when a case of hepatitis A is considered to be infectious is defined as.
Management of HCV-infected patients before and after kidney transplantation S53 Guideline 5. Adults with or exposed to hepatitis C HCV infection. It is estimated that there are 40 million people chronically infected with Hepatitis B Virus HBV and based on some regional level studies it is estimated that there are 6-12 million people with Hepatitis C.
CDC is augmenting previous guidance to recommend. This summary focuses on updated recommendations related to antiviral therapy as of 1 May 2018. A Jaundice OR b Peak elevated total bilirubin levels.
PURPOSE AND OVERVIEW The Federal Bureau of Prisons BOP Clinical Guidance on Evaluation and Management of Chronic Hepatitis C Virus HCV Infection provides the most current BOP recommendations for the treatment of chronic HCV infection in the federal inmate. Clinical care for patients with HCV-related. Seeff4 This document has been approved by the AASLD the Intended for use by physicians these recommenda- Infectious Diseases Society of America and the American tions suggest preferred.
AASLD PRACTICE GUIDELINES Diagnosis Management and Treatment of Hepatitis C. Until recently an. Guidelines for the care and treatment of persons diagnosed with chronic hepatitis c virus infection july 2018 guidelines.
Federal Bureau of Prisons Evaluation and Management of Chronic HCV Infection Clinical Guidance January 2018 1 1. Since the last published document 20 certain previously recommended regimens. 2Hepatitis C diagnosis.
One or more of the following. Hepatitis C screening at least once in a lifetime for all adults aged 18 years and older except in settings where the prevalence of HCV infection is less than 01 Hepatitis C screening for all pregnant women during each pregnancy. Referral treatment care and follow-up of children and adults with or exposed to hepatitis C HCV infection.
The main international guidelines on the management of hepatitis C are listed in Table 1. HCV RNA testing should be performed in Individuals who are anti-HCV positive. Hepatitis C Screening and Follow-Up The identification of risk factors associated with contracting HCV infection served as the basis for the risk-based hepatitis C screening recommenda-tions issued by the US Centers for Disease Control and Prevention CDC in 19988 Although sen-sitive for the identification of persons with chronic.
Hepatitis C Virus Disease Management and Investigative Guidelines Version 012020 Hepatitis C Page 1 CASE DEFINTION CDC 2020 Clinical Criteria for Public Health Surveillance Acute. Recommendations All patients with suspected HCV infection should be tested for anti-HCV antibodies in serum or plasma as first-line diagnostic test A1. An Update Marc G.
Educate the parentsguardians about hepatitis C and how to reduce the risk of transmission. Anti-Hepatitis C virus HCV antibody prevalence in the general population is estimated to be between 009-15. AASLD Diagnosis Management and Treatment of Hepatitis C PDF 2009.
Patients with unexplained liver disease whose anti-HCV is. The main international guidelines on the management of hepatitis C are. Methods for guideline development S86 Appendix 3.
Hepatitis C search strategy. Patients in whom antiviral treatment is being considered. Table 1 International HCV gold standard practice guidelines.
WC 536 World Health Organization 2014 All rights reserved. Liver biopsy in patients with CKD S80 Appendix 2. 1 GUIDELINES FOR THE MANAGEMENT OF HEPATITIS C HEPATITIS VIRAL A brief overview of hepaticviruses other than C - - The hepatitis A virus HAV infection is usually acquired by the fecal-oral route produces a self- limited disease that does not result in chronic infection or long-term liver disease.
ISBN 978 92 4 154875 5 NLM classification. Guidelinesone each for hepatitis A hepatitis B and hepatitis C. The HCV guidance provides peer-reviewed unbiased evidence-based recommendations to aid clinicians with decisions throughout the course of HCV management.
Diagnosis and management of kidney diseases associated with HCV infection S69 S78 Appendix 1. AASLD Update on Treatment of Genotype 1 Chronic Hepatitis C Virus Infection PDF 2011 The American Association of the Study of Liver Diseases has published an IDSA-endorsed practice guideline for the treatment of genotype 1 chronic hepatitis C patients. All persons suspected of having acute or chronic hepatitis C or are at increased risk of HCV infection should be tested for anti-HCV.
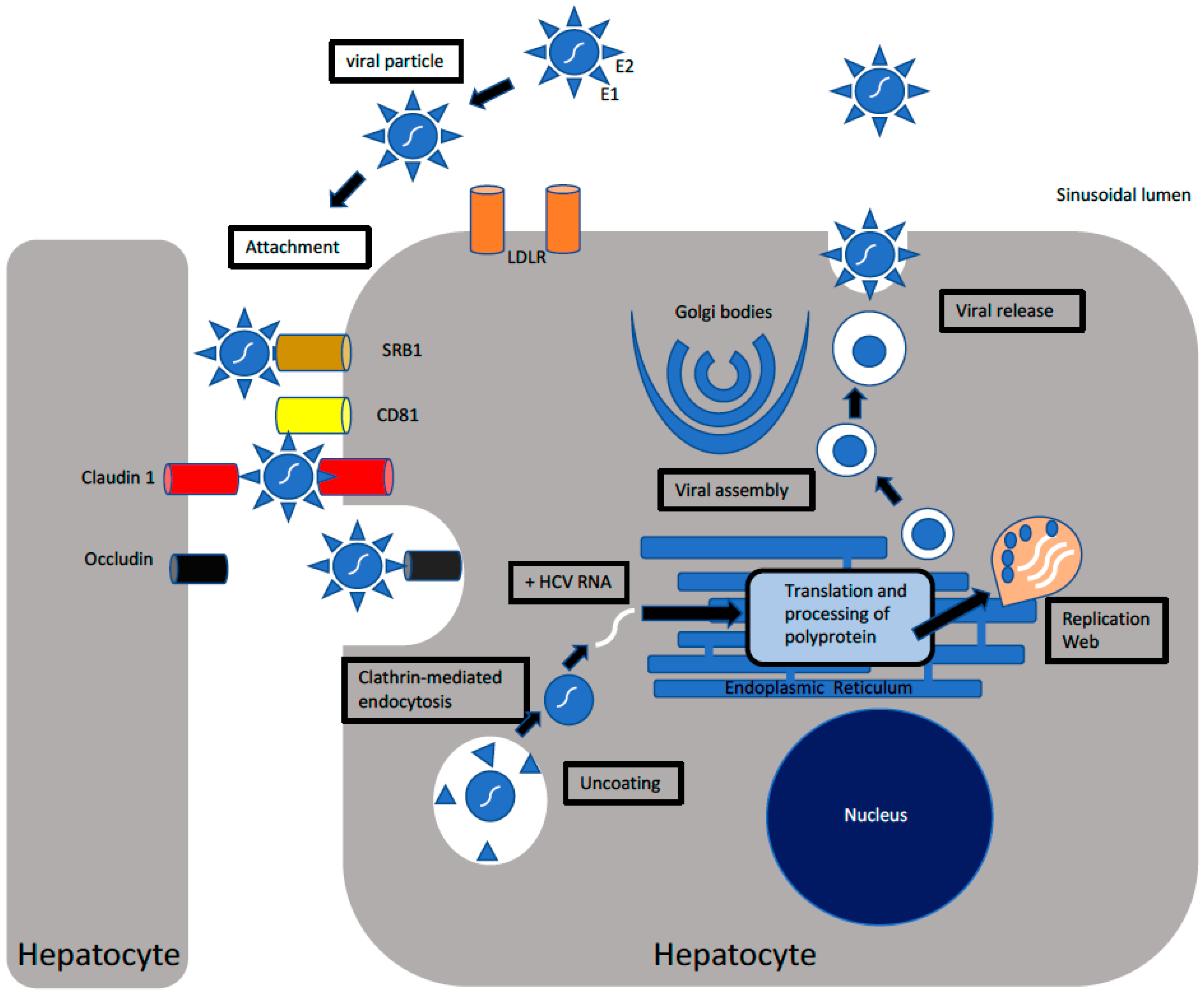
Cells Free Full Text Hepatitis C Virus Infection Host Virus Interaction And Mechanisms Of Viral Persistence Html
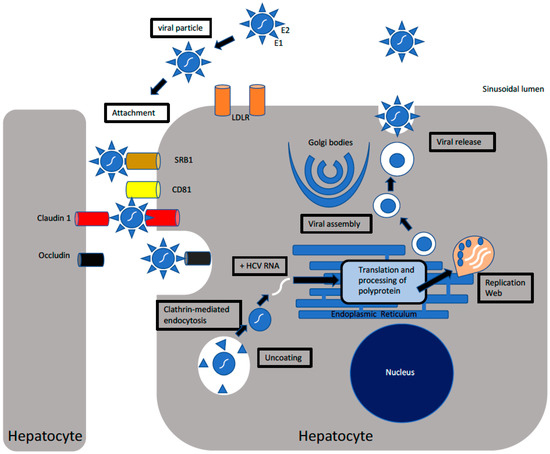
Viruses Free Full Text Regulated Entry Of Hepatitis C Virus Into Hepatocytes Html

Evidence Based Recommendations On The Management Of Extrahepatic Manifestations Of Chronic Hepatitis C Virus Infection Journal Of Hepatology

Cells Free Full Text Hepatitis C Virus Infection Host Virus Interaction And Mechanisms Of Viral Persistence Html

Hepatitis C Virus Trends In Microbiology

2021 Hepatitis C Hep C Hcv Cure Clinical Trials And Research
66 Hepatitis C Workup And Treatment In Primary Care The Curbsiders
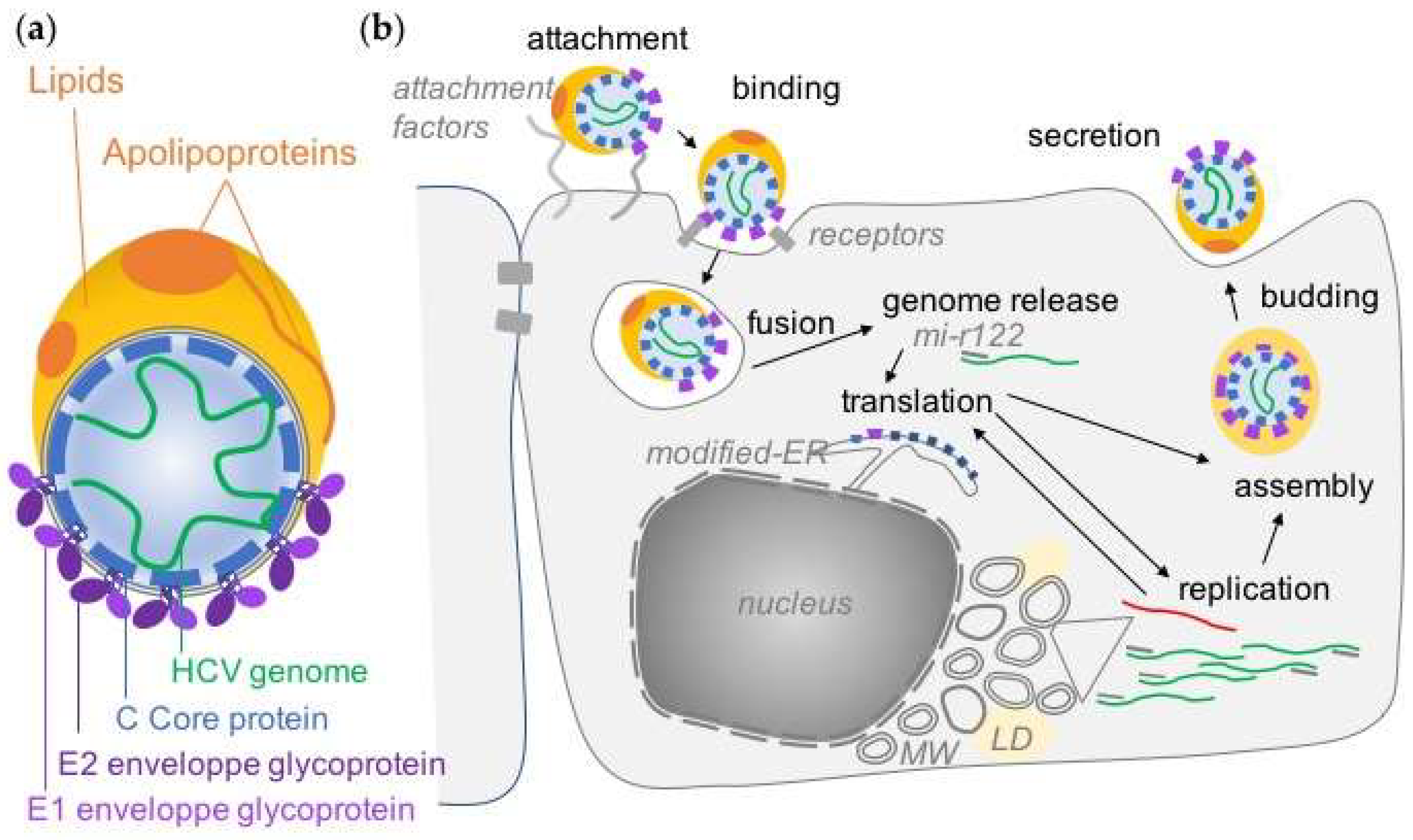
Viruses Free Full Text Overview Of Hcv Life Cycle With A Special Focus On Current And Possible Future Antiviral Targets Html
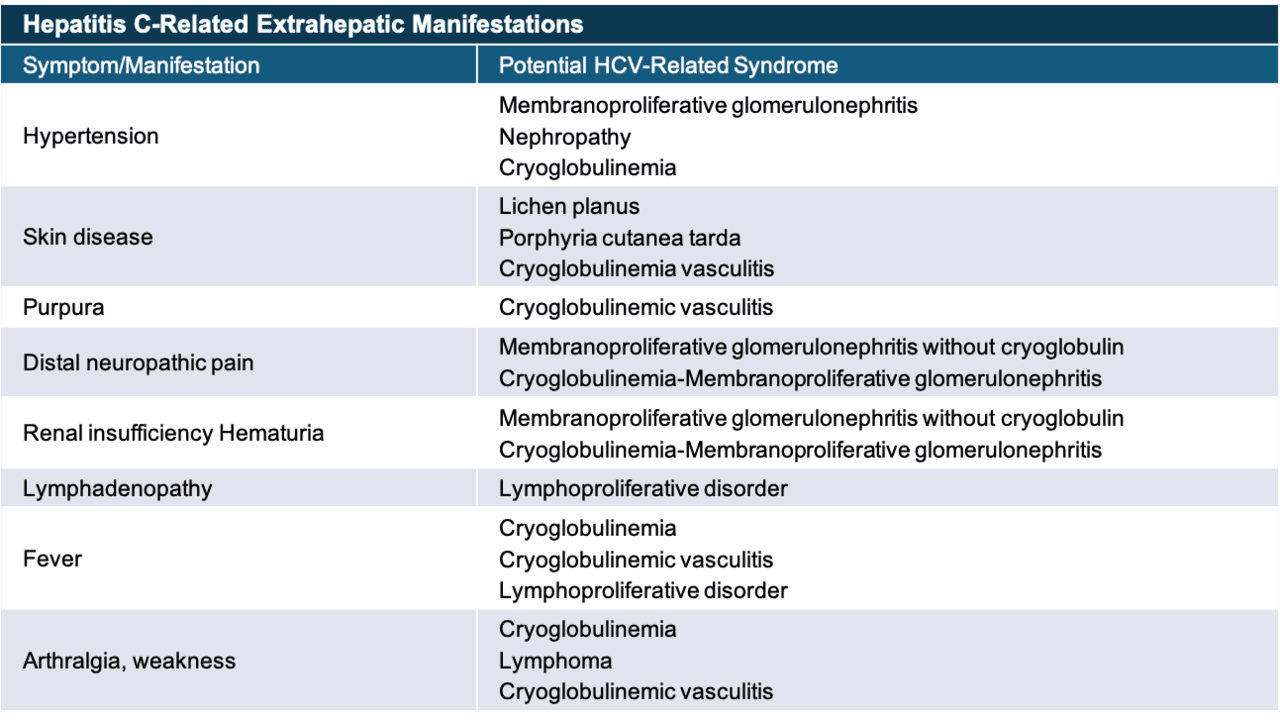
Core Concepts Extrahepatic Conditions Related To Hcv Infection Evaluation Staging And Monitoring Of Chronic Hepatitis C Hepatitis C Online

Meeting The Who Hepatitis C Virus Elimination Goal Review Of Treatment In Paediatrics Kim 2020 Journal Of Viral Hepatitis Wiley Online Library

Effects And Cost Of Different Strategies To Eliminate Hepatitis C Virus Transmission In Pakistan A Modelling Analysis The Lancet Global Health

Interim Effect Evaluation Of The Hepatitis C Elimination Programme In Georgia A Modelling Study The Lancet Global Health
Hcv Extrahepatic Manifestations
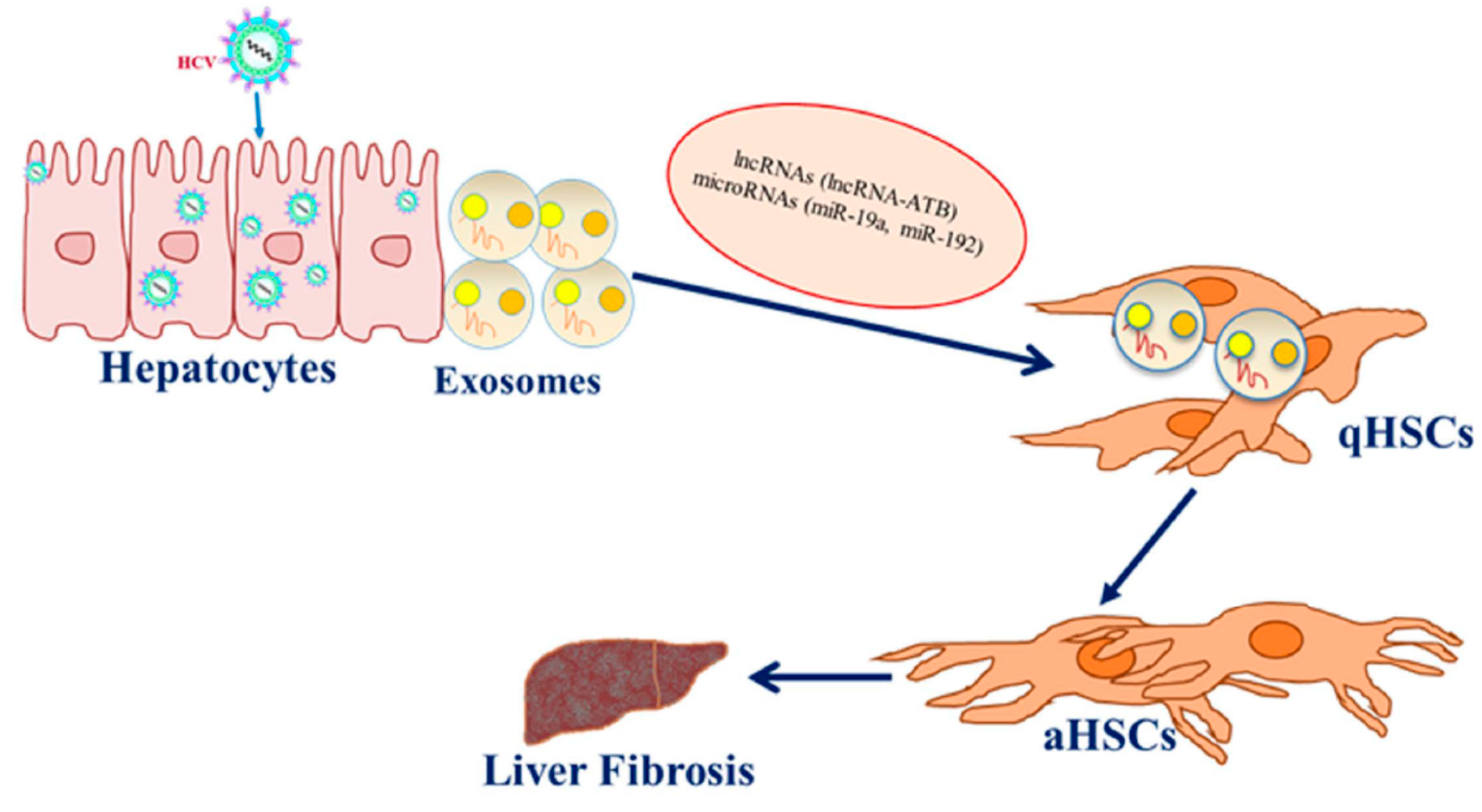
Cells Free Full Text Mechanisms Underlying Hepatitis C Virus Associated Hepatic Fibrosis Html

In A Critical State Ongoing Barriers To Treatment For Hepatitis C Virus Hcv The American Journal Of Medicine

Hepatitis C Virus Life Cycle In Cells Infection And Host Response And Analysis Of Molecular Markers Influencing The Outcome Of Infection And Response To Therapy Clinical Microbiology And Infection
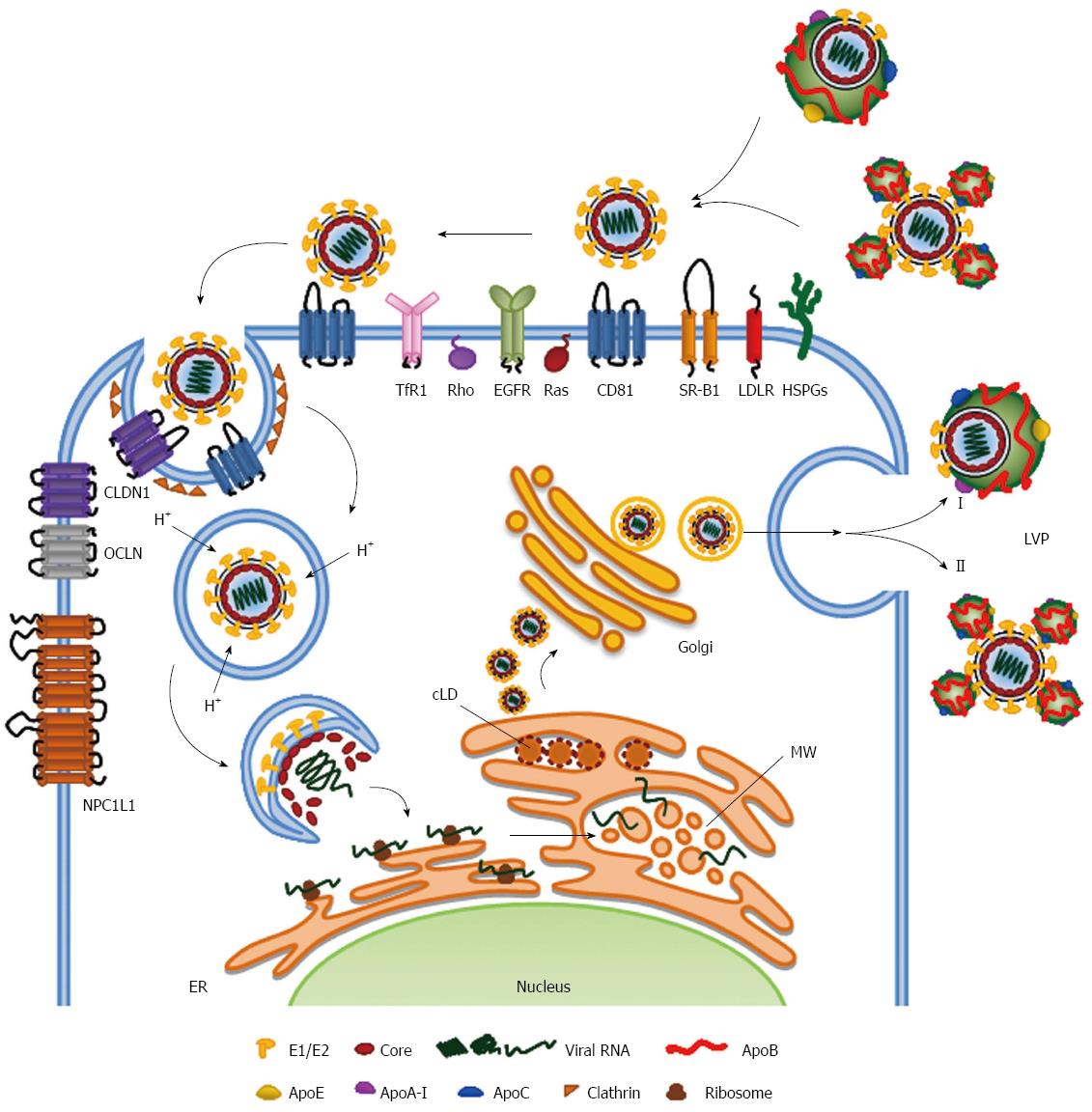
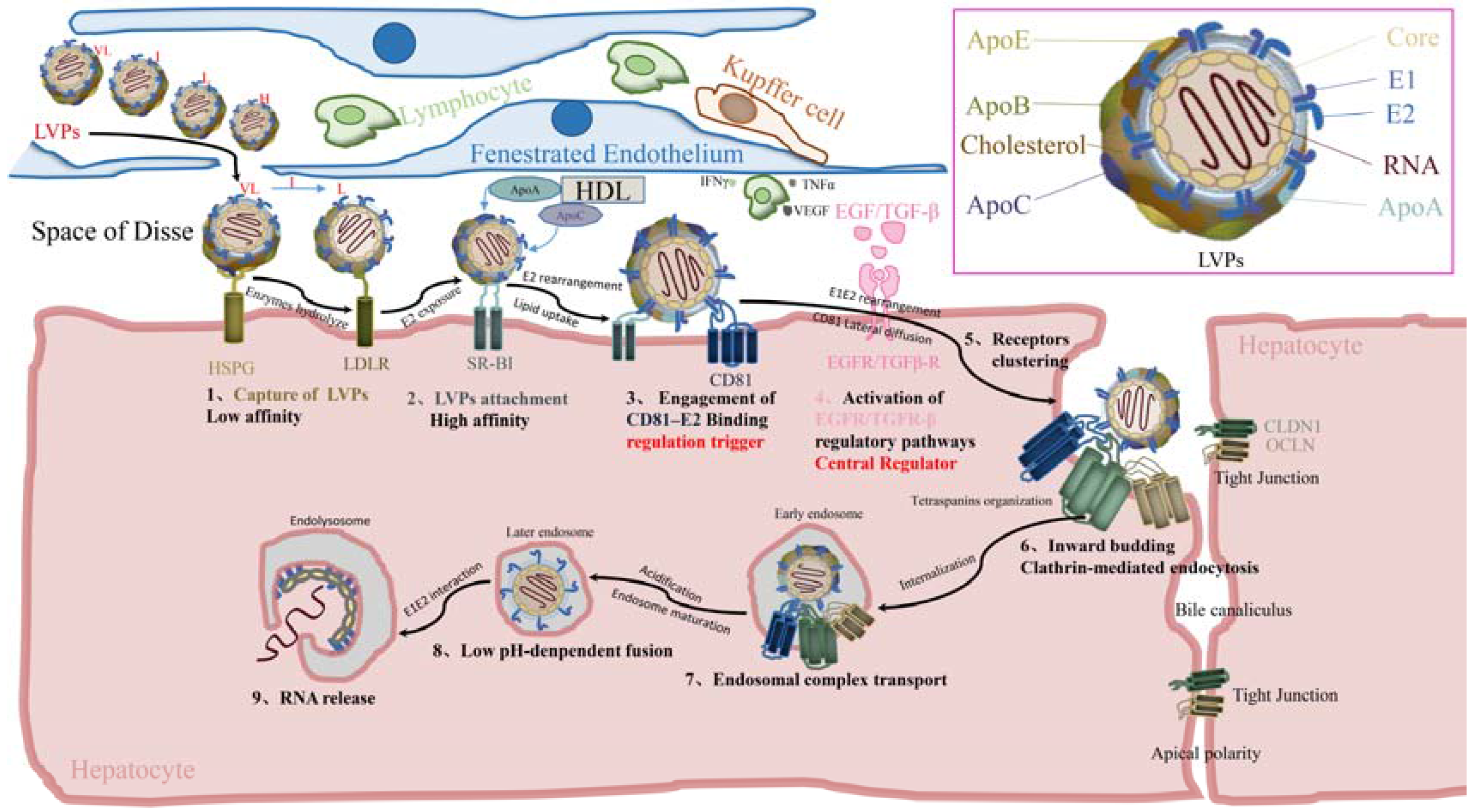
Hepatitis C Virus Relies On Lipoproteins For Its Life Cycle
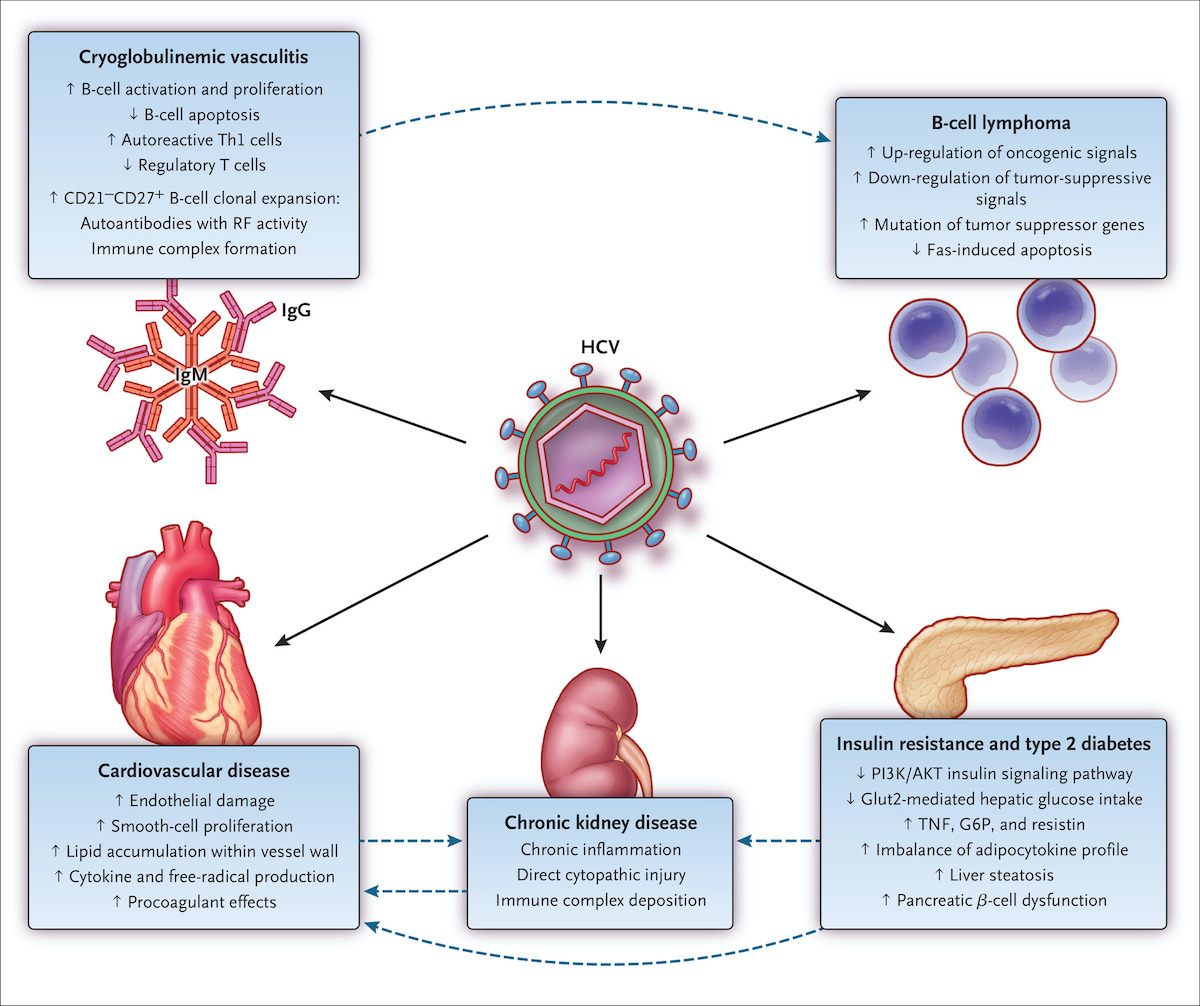
Core Concepts Extrahepatic Conditions Related To Hcv Infection Evaluation Staging And Monitoring Of Chronic Hepatitis C Hepatitis C Online
